Surgical site infections
Healthcare-associated infections (HAIs) are infections that occur while receiving health care. Patients with medical devices (central lines, urinary catheters, ventilators) or who undergo surgical procedures are at risk of acquiring HAIs. HAIs continue to be a tremendous issue today. Surgical site infections (SSIs) are the most common healthcare-associated infections among surgical patients. It is obviously important to improve patient safety by reducing the occurrence of surgical site infections. SSIs remain a major clinical problem in terms of morbidity, mortality, length of hospital stay and overall direct and not-direct costs in all regions of the world. Despite progress in prevention knowledge SSIs remain one of the most common adverse events in the surgical departments worldwide.
Surgical site infections are infections that occur after surgery in the part of the body where the surgery took place. Surgical site infections can sometimes be superficial infections involving the skin only. Other surgical site infections are more serious and can involve tissues under the skin, organs, or implanted material. The Centers for Disease Control and Prevention (CDC) has defined SSIs to standardize data collection for the National Nosocomial Infections Surveillance (NNIS) program. SSIs are classified into incisional SSIs, which can be superficial or deep, and organ/space SSIs, which affect the rest of the body other than the body wall layers (see the image below). These classifications are defined as follows:
Superficial incisional SSI – Infection involves only skin and subcutaneous tissue of incision
Deep incisional SSI – Infection involves deep tissues, such as fascial and muscle layers; this also includes infection involving both superficial and deep incision sites and organ/space SSI draining through incision
Organ/space SSI – Infection involves any part of the anatomy in organs and spaces other than the incision, which was opened or manipulated during operation.
Preventing SSIs is a global priority. Bacteria are becoming increasingly resistant to antibiotics, making SSIs prevention even more important nowadays. SSIs prevention is complex and requires the integration of a range of measures before, during, and after surgery. Both the World Health Organization (WHO) and the Centers for Disease Control and Prevention (CDC) have recently published guidelines for the prevention of SSIs.
WHO Global guidelines for the prevention of surgical site infection
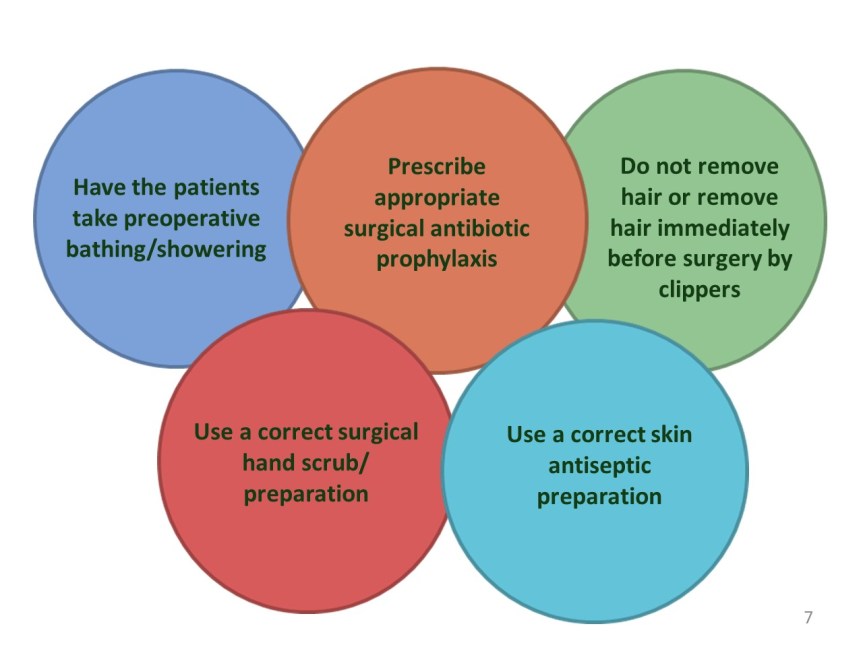
The 2016 WHO Global guidelines for the prevention of surgical site infection are evidence-based including systematic reviews presenting additional information in support of actions to improve practice. The guidelines include 13 recommendations for the pre-operative period, and 16 for preventing infections during and after surgery. They range from simple precautions such as ensuring that patients bathe or shower before surgery, appropriate way for surgical teams to clean their hands, guidance on when to use prophylactic antibiotics, which disinfectants to use before incision, and which sutures to use. The proposed recommendations are as follows:
“Strong” – Expert panel was confident that benefits outweighed risks, considered to be adaptable for implementation in most (if not all) situations, and patients should receive intervention as course of action.
“Conditional” – Expert panel considered that benefits of intervention probably outweighed the risks; a more structured decision-making process should be undertaken, based on stakeholder consultation and involvement of patients and healthcare professionals.
Surgical care bundles
Numerous clinical interventions with varying levels of supporting evidence have been implemented to reduce SSIs among surgical patients. A recent approach to improving patient outcomes is the use of care bundles. Care bundles were first introduced by the Institute for Healthcare Improvement (IHI) in 2001 to improve clinical outcomes in the critical care population. The concept of a care bundle was developed from evidence documenting that a structured approach to performing 3–5 evidence-based collective interventions could lead to an improved patient outcome. While specific interventions may vary between bundles, it is the bundle approach that ensures consistent implementation of all measures that is claimed to be successful. Surgical care bundles have been developed to reduce SSIs. A proposal for a bundle for the prevention of surgical site infections worldwide.
Surgical antibiotic prophylaxis
Antibiotic prophylaxis (AP) is one of the most important component of a perioperative infection prevention strategy. The use of AP contributes considerably to the total amount of antibiotics used in hospitals and may be associated with increases in antibiotic resistance and healthcare costs. Although AP plays a pivotal role in reducing the rate of surgical site infections, other factors such as attention to basic infection-control strategies may have a strong impact on surgical site infections rates. Perioperative surgical AP should be recommended for operative procedures that have a high rate of postoperative wound infection or when foreign material is implanted. Prophylactic antibiotic agents should be nontoxic and inexpensive and have in vitro activity against the common organisms that cause postoperative wound infection after a specific surgical procedure. Therapeutic concentrations of antibiotics should be present in the tissue throughout the all period that the wound is open.
Principles of appropriate antibiotic prophylaxis
1. Antibiotic agents alone are unable to prevent surgical site infections. Strategies to prevent surgical site infections should always include attention to:
- IPC strategies including correct and compliant hand hygiene practices
- Meticulous surgical techniques and minimization of tissue trauma
- Hospital and operating room environments
- Instrument sterilization processes
- Peri-operative optimization of patient risk factors
- Peri-operative temperature, fluid, and oxygenation management
- Targeted glycemic control
- Appropriate management of surgical wounds
2. Antibiotic prophylaxis should be administered for operative procedures that have a high rate of post-operative surgical site infection, or when foreign materials are implanted.
3. Antibiotic agents given as prophylaxis should be effective against the aerobic and anaerobic pathogens most likely to contaminate the surgical site—i.e., gram-positive skin commensals or normal flora colonizing the incised mucosae.
4. Antibiotic prophylaxis should be administered within 120 minutes before the incision. Administration of the first dose of antibiotic agents beginning within 30 to 60 minutes before surgical incision, however, is recommended for most antibiotic agents (e.g., cefazolin), to ensure adequate serum and tissue concentrations during the period of potential contamination. Obese patients ≥120 kg require higher doses of antibiotic agents.
5. A single dose generally is sufficient. Additional antibiotic doses should be administered intra-operatively for procedures >2–4 hours (typically where duration exceeds two half-lives of the antibiotic) or with associated significant blood loss (>1.5 L).
6. There is no evidence to support the use of post-operative antibiotic prophylaxis.
7. Each institution is encouraged to develop guidelines for proper surgical prophylaxis.
Hospital-based programs dedicated to improving antibiotic use, commonly referred to as Antimicrobial Stewardship Programs (ASPs), can both optimize the management of infections and reduce adverse events associated with antibiotic use. Therefore, every hospital worldwide should utilize existing resources to create an effective multi-disciplinary team. The preferred means of improving antibiotic stewardship should involve a comprehensive program that incorporates collaboration between various specialties within a healthcare institution including infectious disease specialists, hospital pharmacists, clinical pharmacologists, administrators, epidemiologists, infection prevention and control specialists, microbiologists, surgeons, anesthesiologists, intensivists, and underutilized but pivotal stewardship team members-the surgical, anesthetic, and intensive care nurses in our hospitals.
The ASP policies should be based on both international and national antibiotic guidelines and tailored to local microbiology and resistance patterns. Facility-specific treatment recommendations, based on guidelines and local formulary options promoted by the APS team, can guide clinicians in antibiotic agent selection and duration for the most common indications for antibiotic use. Standardizing a shared protocol of antibiotic prophylaxis should represent the first step of any ASP.